A Comparative Analysis of Sugar Cane and Sugar Beet Fermentation
Raymond Jagessar*1, Alexica Brown2
1Department of Chemistry, Faculty of Natural Sciences, University of Guyana
2Final Year Chemistry student, Faculty of Natural Sciences, University of Guyana
Received Date: April 22, 2025; Accepted Date: April 30, 2025; Published Date: July 19, 2025;
*Corresponding author: Raymond Jagessar, Department of Chemistry, Faculty of Natural Sciences, University of Guyana; Email: raymond.jagessar@uog.edu.gy
Citation: Jagessar R, Brown A (2025); A Comparative Analysis of Sugar Cane and Sugar Beet Fermentation; Enviro Sci Poll Res and Mang: ESPRM-174
DOI: 10.37722/ESPRAM.2025201
Abstract
With a view to curb global warming, it is required that countries pursue a net zero emission of carbon dioxide. CO2 comes mainly from the burning of fossil fuel. To this effect, alternative energy sources such as bioethanol must be sought. Two feedstocks are sugar cane and beet fruit. The latter is not a sustainable feedstock, whereas sugar cane is. These were fermented to produce bioethanol using the biological catalyst Saccharomyces cervisae. The sugar cane demonstrated a high initial average Brix content of 18.97, compared to the average Brix content of 7.87 for beet fruit. The % reduction of the Brix content seems to be the same in both instances (72%). The ethanolic content of the distillates were analysed using Gas Chromatography, GC. The highest average % yield of bio ethanol (10.18%) was noted for the sugar cane juice, whereas a value of 4.1 % was observed for sugar beet. The Gas Chromatograph for both substrates also indicated the presence of other minor components such as acetone, methylacetate, propanol, ethylacetate, butan-2-ol, isobutanol, butanol, acetal, pentanol and furfuraldehyde. The sugar beet GC was much cleaner with side products: propanol, ethylacetate and isobutanol. This demonstrated that sugar cane is a far superior source for bioethanol, compared with sugar beet.
Keywords: Bioethanol production, Sugar cane, Beet, Fermentation, Environmental impact
Information Board
ISSN: 2641-6816
Frequency: Continuous
Format: Video, PDF and HTML
Versions: Online (Open Access)
Language: English
Impact Factor: 4.21
Quick Links
Issues
Video Gallery
Guidelines
Publication Fee
Editorial Board
Review Board
Publish your article not just in PDF and HTML, but also in documentary format. Simply submit your manuscript as usual — we’ll take care of the rest.
Introduction
With a view to decrease dependence on fossil fuel, as a result of depletion, increasing global fuel price, increasing population and increasing global warming, there has been increased interest in the use of renewable energy sources of which bioethanol is one [1,2,3]. Bioethanol (b.p: 78.5°C) can be used for a variety of purposes, of which blending with gasoline to produce gas alcohol to power automobiles is of current utilisation [1,2,3, 24-25]. In addition, ethanol is a clean burning renewable energy source4. Ethanol is also an important component of alcoholic beverages such as wine, beer, cider, vodka, gin. whisky, brandy etc. It is also an important starting materials for aldehydes, ketones, carboxylic acid, carboxylic acid derivatives and the hydroxyl group is a component of many pharmaceutical drugs 5. Ethanol can be used in the perfume, disinfectant, tincture, biological and biofuel industries. Ethanol production through Fermentation has been one of the world most significant approaches to aid in the Advancement of Commercial Industry.
Ethanol doesn’t have significant environmental impact as fossil fuel combustion [3]. It has low air polluting effect and low atmospheric photochemical reactivity, further reducing impact on the ozone layer [6]. It contributes little net CO2 accumulation to the atmosphere and thus should curb global warming6-9.
Ethanol can be used in three primary ways as biofuel, namely, E10 which is a blend of 10% ethanol and 90% unleaded gasoline, a component of reformulated gasoline both directly and or as ethyl tertiary butyl ether (ETBE) and as E85 which is 85% ethanol and 15% unleaded gasoline. When mixed with unleaded gasoline, ethanol increases octane levels, decreases exhaust emissions and extends the supply of gasoline. Bio-ethanol is made by fermenting almost any material that contain starch or sugar. Grains such as corn and sorghum are good sources, but fruits that are high in sugar concentration are excellent sources as well, since they contain ready to ferment sugars [10].
To solve the above problem, emanating from fossil fuel, one alternative is to produce bioethanol from fruits, other grown organic matter or waste [3,4,6-8,9]. Bioethanol can be obtained via the fermentation of glucose, fructose or sucrose under the influence of Saccharomyces cerevisiae at room temperature, [4,6-35]. This is shown in Figure 1. Also, acid hydrolysis of lignocellulose material followed by subsequent fermentation [7-21]. Sugar rich sources include ripe fruits [8-18] etc. Other sources include biodegradable fraction of products, waste and residues from agriculture like vegetables, fruit wastes and animal origin [11-12, 17, 20-21] etc. The percentage yield of ethanol, ranging from 4.0 -10.0 v/v) have been reported [3-12]. Fruits that are high in sugar concentration are favourable to the fermentation process, since they can produce high percentage volume of ethanol
Fermentation is the process of energy production in a cell in an anerobic environment with the lack of an external electron acceptor [22]. Sugars are the common substrate of fermentation and the products include ethanol, lactic acid and hydrogen. In some instances, compounds such as butyric acid and acetone are produced.
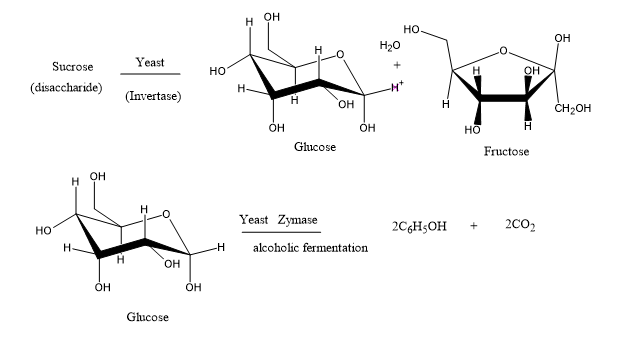
Figure 1: Fermentation route of glucose and sucrose
The fermentation process begins with the yeast breaking down the different forms of sugar in any fermenting matrix. Saccharomyces cerevisiae contains two enzymes that is very important for the yeast enzyme activity in the fermentation process. These two enzymes are called Invertase and Zymase and they functions are similar but somewhat prerequisite to each other. Invertase aids in converting any sucrose sugar that is present in any biomass that is used in fermentation to glucose and fructose while zymase aids in the conversion of glucose to ethanol [22]., Figure 1.
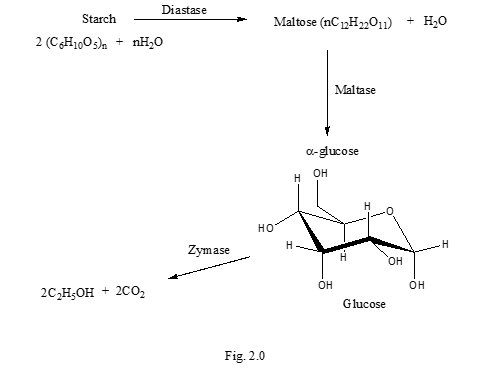
During Fermentation, starch is first hydrolysed to maltose by the action of the enzyme diastase. This enzyme is obtained from germinating barley seeds or malt. Maltose is converted to glucose by the enzyme maltase. Glucose is then fermented to ethanol via the enzyme zymase [23], Figure 2. Once the sugars are broken down into monosaccharides, the yeast can now use them. Saccharomyces cerevisiae is able to perform both aerobic and anaerobic respiration.
Twenty research articles were reviewed, with the view to identify research gaps and several points can be noted [24-63]. Several feedstocks were reported and these are beet molasses, sugar cane, sugar beet molasses, sugar beet syrup, sugar beet pulp, beet root wine, beet juice, sugar beet. To the best of knowledge, only one article reported the fermentation of sugar cane vs beet sugar in the production of sparkling wines. Sixteen of these articles reported the use of Saccharomyces cervisae. One article reported the use of L. acidophilus, L. plantarum, and scheffersomyces stipites, Aspergillus niger was reported as biological agent to convert sugar rich feedstock above to bioethanol. Varying % yield for bioethanol were obtained. No experimental design was mentioned for twelve of these articles. For ten of these articles, no statistical analyses was used. ANOVA was the dominant statistical tool to test for significance difference. For one article, the Duncan T-test was employed. For another article, Linear Regression and T-tests were used to analyse results. One of the experimental design employed a 2 x 2 Factorial design with two factors: sugar type and time points.
The objectives of this research were to compare the fermentation yield of sugar cane versus sugar beet. The rationale for using these two substrates is that sugar cane is an abundant sustainable feedstock in the Caribbean. In addition, sugar beet can be grown in certain parts of Guyana. Sugar beet is also an alternative sweetener in the Europe and its fermentation profile must be evaluated. This has made it necessary to compare the ethanolic fermentation yield of both sugar rich substrates. These information will dictate the further expansion of the sugar cane industry in Guyana and the rest of the Caribbean.
Methodology
Experimental Design:
The experimental design used was the Completely Randomised Block Design (CRD). The three treatments were replicated three times. There were a total of 6 experimental units
Sugar Cane Method:
The process began by obtaining mature sugar cane stalks from three different sources. Care was taken to ensure the sugar cane was healthy and free from diseases or pests. Following harvesting, the juice was extracted from the sugar cane stalks using a mechanical mill. The sugar cane was randomly selected. A refractometer was used to measure the initial sugar content (°Brix) of the sugar cane juice.
Experiments were conducted in triplicates. The jars were collected and labelled Jar 1, Jar 2, and Jar 3. 500 ml of sugar cane juice was added to each of the jars. The initial pH was measured and citric acid was added to each medium to lower the pH. This is because the organisms, specifically yeast, which produce the enzymes needed for glucose fermentation, have adapted to thrive in acidic environments (Yalcin & Ozbas, 2008).
For the fermentation process, the jars were closed, and tubes were connected to the jars leading to a test tube with paraffin. During fermentation, microorganisms consumed the sugars in the sugar cane juice and produced carbon dioxide (CO₂) as a byproduct (Pepin & Marzzacco, 2021). The gas needed to escape from the jar to prevent pressure buildup. The tube allowed the CO₂ to bubble out into the paraffin solution.
After 72 hours, the mixture was filtered. The filtrate was obtained and collected in 500 ml water bottles. The Brix of each filtrate sample was measured. The filtrate was distilled using simple distillation at a temperature range of 80°C to 95°C. For each jar, six fractions were collected and added to six test tubes. The filtrate’s composition was tested for the percentage of alcohol using a pictometer. The ethanol concentration and other components were analysed using gas chromatography. The ethanol standard used had a 40% ethanol concentration and 3.775 g/HL for the 14 analytes.
Sugar Beet Method:
The beet was washed with distilled water, dried, weighed, and cut into smaller pieces. Experiments were conducted in triplicates. Drying the beet removed excess water, providing a more accurate measurement of the actual biomass. Blending the beet into smaller pieces increased the surface area available for enzymatic and microbial activity during fermentation, enhancing the efficiency of the process. 200 g of beetroot was weighed. The determination of the volume of beet was conducted using a graduated measuring cylinder. The beet was introduced into a cylinder filled with 40 ml of water. The volume difference observed before and after the complete immersion of the feedstock represented its volume (Vf). The specific gravity of the feedstock was computed as the ratio of the feedstock’s mass (Mf) to its volume (Vf). Specific gravity holds significance in fermentation studies, aiding in the assessment of feedstock density, providing insights into its composition, and predicting fermentation efficiency.
The jars were collected and labelled Jar 1, Jar 2, and Jar 3. 605 g of beetroot was blended with 500 ml of water and added to each of the jars. For the fermentation process, a solution of inoculated yeast, Saccharomyces cerevisiae, was added to the medium. After 72 hours, the mixture was filtered. The filtrate was obtained and collected in 500 ml water bottles. The Brix of each filtrate sample was measured for Jar 1, Jar 2, and Jar 3. The filtrate was distilled using simple distillation at a temperature range of 90°C to 98°C. For each jar, six fractions were collected and added to six test tubes. The filtrate’s composition was tested for the percentage of alcohol using a pictometer. The ethanol concentration and further analysis were done on the sample using gas chromatography. The ethanol standard used had a 40% ethanol concentration and 3.775 g/HL for the 14 analytes.
Preparation of 12% Yeast Solution:
To prepare the 12% yeast solution, 44 ml of deionized water was added to a 100 ml beaker, and then 6 g of dry yeast (Saccharomyces cerevisiae) were introduced. Two brands of yeast were used at equal mass, Fermipan and Pakmaya. The mixture was allowed to incubate for 90 minutes at 27°C to attain a yeast solution with a concentration of 12%.
GC Analyses:
The ethanol concentration and Gas chromatography analysis were done at the Central Laboratory at the Quality Assurance Department at Demerara Distillers Ltd.
Results
Table 1. Fermentation of Sugar cane juice
| Cane Juice | Initial o Brix | Final o Brix | Initial pH | Final pH | Weight of Residue | Final Specific Gravity | Average Ethanol Content % |
| Jar 1 | 19.00 | 5.20 | 6.17 | 2.91 | 1.737 | 1.00 | 9.86 |
| Jar 2 | 19.10 | 5.40 | 5.35 | 3.29 | 4.683 | 1.08 | 10.65 |
| Jar 3 | 18.80 | 4.90 | 4.96 | 4.42 | 3.221 | 1.02 | 10.03 |
| Average | 18.967 | 5.167 | 5.493 | 3.540 | 3.214 | 1.033 | 10.181 |
Table 2. Fermentation of Sugar beet
| Sugar beet | Initial o Brix | Final o Brix | Initial pH | Final pH | Weight of Residue | Final Specific Gravity | Average Ethanol Content % |
| Jar 1 | 7.90 | 2.00 | 5.91 | 3.17 | 529.44 | 1.00 | 3.89 |
| Jar 2 | 7.80 | 2.50 | 5.86 | 2.74 | 516.84 | 1.08 | 4.935 |
| Jar 3 | 7.90 | 2.10 | 5.89 | 3.19 | 493.84 | 1.02 | 3.47 |
| Average | 7.867 | 2.200 | 5.887 | 3.033 | 513.37 | 1.033 | 4.103 |
Table 3: Showing the ethanol content % for Sugar cane and sugar beet in the various fractions
| Fractions | Ethanol Content % | ||||
| Jar 1 | Jar 2 | Jar 3 | Average | ||
| Sugar cane | 1 | 35.01 | 58.17 | 32.65 | 41.94 |
| 2 | 3.58 | 0.18 | 18.48 | 7.41 | |
| 3 | 1.56 | 1.46 | 8.2 | 3.74 | |
| 4 | 11.35 | 0 | 0.11 | 3.82 | |
| 5 | 7.48 | 4.11 | 0.73 | 12.32 | |
| 6 | 0.18 | 0 | 0 | 0.06 | |
| Sugar beet | 1 | 11.72 | 12.44 | 11.29 | 11.82 |
| 2 | 10.86 | 12 | 8.81 | 10.56 | |
| 3 | 0.23 | 4.49 | 0.2 | 1.64 | |
| 4 | 0.26 | 0.68 | 0.29 | 0.41 | |
| 5 | 0.13 | 0 | 0.26 | 0.13 | |
| 6 | 0.13 | 0 | 0.07 | 0.07 | |
Table 4: Fermentation Profile for Sugar Cane vs. Sugar Beet
| Components | Sugar Cane (G/HL) | Sugar Beet (G/HL) |
| Acetaldehyde | (+) | (+) |
| Methanol | (+) | (+) |
| Acetone | (+) | (-) |
| 1 Methylacetate | (+) | (-) |
| 2 Propanol | (+) | (+) |
| 1 Ethylacetate | (+) | (+) |
| 2 Butan-2 ol | (+) | (-) |
| 2 Isobutanol | (+) | (+) |
| 2 Butan-1 ol | (+) | (-) |
| Acetal | (+) | (-) |
| 2 Isoamyl Alcohol | (-) | |
| 2 Activeamyl Alcohol | (-) | |
| 2 1 pentanol | -(+) | (-) |
| Furfuraldehyde | (+) | (-) |
Figure 3: Showing the results for GC analysis for the ethanol standard
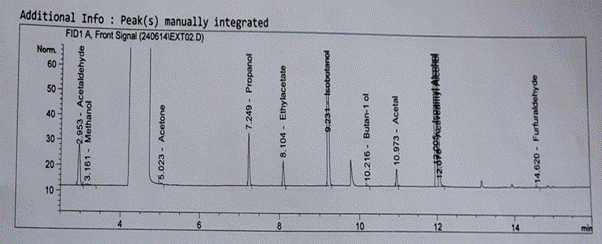
Figure 4: Showing the results for GC analysis for sugar cane sample

Figure 5. Showing the results for GC analysis for the sugar beet sample
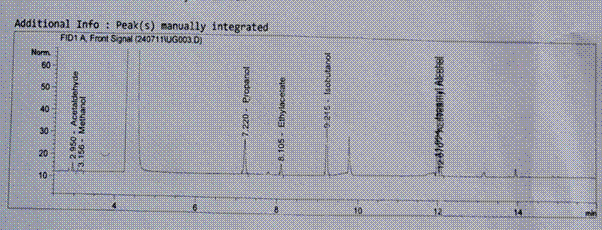
Figure 6: A Plot of Initial Brix vs Final Brix per Feedstock
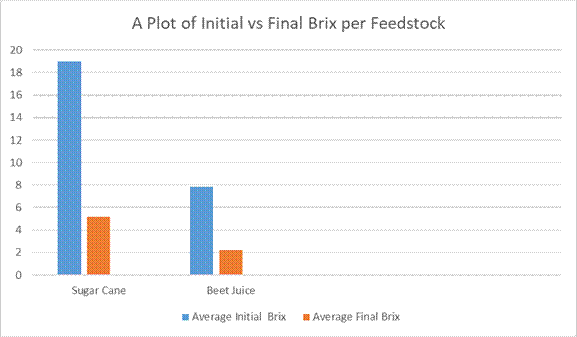
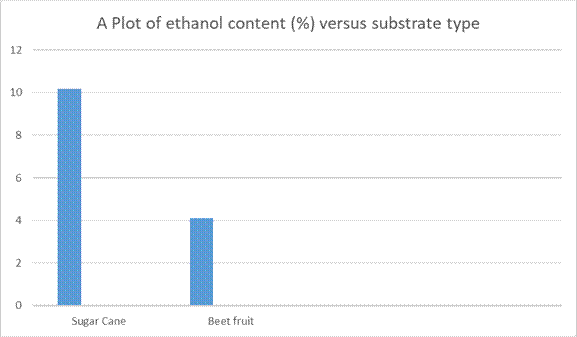
Figure 7: A Plot of ethanol content versus feedstock type
Analysis of Results
Statistical analysis will be performed using Microsoft Excel 365, employing Analysis of Variance (ANOVA) at a 95% confidence interval to analyse performance data.
Table 5: Anova: Two-Factor with Replication
| SUMMARY | Jar 1 | Jar 2 | Jar 3 | Total | ||
| Sugar cane | ||||||
| Count | 6 | 6 | 6 | 18 | ||
| Sum | 59.16 | 63.92 | 60.17 | 183.25 | ||
| Average | 9.86 | 10.65333 | 10.02833 | 10.18056 | ||
| Variance | 168.4876 | 544.3688 | 174.3826 | 261.076 | ||
| Sugar beet | ||||||
| Count | 6 | 6 | 6 | 18 | ||
| Sum | 23.33 | 29.61 | 20.92 | 73.86 | ||
| Average | 3.888333 | 4.935 | 3.486667 | 4.103333 | ||
| Variance | 32.9475 | 34.65015 | 26.46715 | 28.06076 | ||
| Total | ||||||
| Count | 12 | 12 | 12 | |||
| Sum | 82.49 | 93.53 | 81.09 | |||
| Average | 6.874167 | 7.794167 | 6.7575 | |||
| Variance | 101.2871 | 272.1084 | 102.9663 | |||
| ANOVA | ||||||
| Source of Variation | SS | df | MS | F | P-value | F crit |
| Sample | 332.3937 | 1 | 332.3937 | 2.03236 | 0.164304 | 4.170877 |
| Columns | 7.738756 | 2 | 3.869378 | 0.023659 | 0.976637 | 3.31583 |
| Interaction | 1.066956 | 2 | 0.533478 | 0.003262 | 0.996744 | 3.31583 |
| Within | 4906.519 | 30 | 163.5506 | |||
| Total | 5247.718 | 35 |
Table 6: Anova: Two Factor without Replication, a comparison of the average % mean of ethanol amongst the different fractions (1-6)
| Anova: Two-Factor Without Replication | ||||||
| SUMMARY | Count | Sum | Average | Variance | ||
| Row 1 | 2 | 53.76 | 26.88 | 453.6072 | ||
| Row 2 | 2 | 17.97 | 8.985 | 4.96125 | ||
| Row 3 | 2 | 5.38 | 2.69 | 2.205 | ||
| Row 4 | 2 | 4.23 | 2.115 | 5.81405 | ||
| Row 5 | 2 | 12.45 | 6.225 | 74.29805 | ||
| Row 6 | 2 | 0.13 | 0.065 | 5E-05 | ||
| Column 1 | 6 | 69.29 | 11.54833 | 238.8107 | ||
| Column 2 | 6 | 24.63 | 4.105 | 30.59987 | ||
| ANOVA | ||||||
| Source of Variation | SS | df | MS | F | P-value | F crit |
| Rows | 972.3771 | 5 | 194.4754 | 2.595248 | 0.15932 | 5.050329 |
| Columns | 166.2096 | 1 | 166.2096 | 2.218045 | 0.196584 | 6.607891 |
| Error | 374.676 | 5 | 74.93519 | |||
| Total | 1513.263 | 11 | ||||
A comparison of the average % mean of ethanol amongst the major fractions (1-2)
Table 7: Anova: Two-Factor Without Replication
| SUMMARY | Count | Sum | Average | Variance | ||
| Row 1 | 2 | 53.76 | 26.88 | 453.6072 | ||
| Row 2 | 2 | 17.97 | 8.985 | 4.96125 | ||
| Column 1 | 2 | 49.35 | 24.675 | 596.1605 | ||
| Column 2 | 2 | 22.38 | 11.19 | 0.7938 | ||
| ANOVA | ||||||
| Source of Variation | SS | df | MS | F | P-value | F crit |
| Rows | 320.231025 | 1 | 320.231 | 1.157225 | 0.47678 | 161.4476 |
| Columns | 181.845225 | 1 | 181.8452 | 0.657138 | 0.566338 | 161.4476 |
| Error | 276.723225 | 1 | 276.7232 | |||
| Total | 778.799475 | 3 |
Discussion
Table 1.0 shows the fermentation data for the sugar cane juice. Of significance, the average inititial Brix decreases from 18.967 to 5.1667 (i.e 72.75% reduction) at the 72 hours stipulated fermentation period. This probably indicated that more yeast were needed for the fermentation process. Brix is a measure of the sugar content in a solution*. This probably indicated that more yeast were needed for the fermentation process. The average ethanol content was found to be 10.18%. There was also a decrease in pH from 5.493 to 3.54. This decrease in pH was probably due to acetic acid formation via oxidation of ethanol. Table 2.0 shows the fermentation profile for sugar beet. It can be seen that the average Brix decrease from 7.867 to 2.20 i.e 72.03% reduction. The pH also decreased from 5.887 to 3.03. This is probably due again to acetic acid formation. The average ethanol content for sugar beet was 4.103 which is lower than that for sugar cane (10.18) = 40.36%. This shows that the sugar cane juice is more fermentable than sugar beet. Table 1.0 and Table 2.0 show that sugar cane juice has a higher average initial Brix content (18.97) than sugar beet (7.87), indicating that it’s a far better feedstock for bioethanol production, compared with sugar beet. However, the sugar substrate conversion efficiency to bioethanol seems to be the same for both, 73 %. .
The initial ethanol concentrations fractions are significantly higher in the sugarcane samples (41.9 %) compared to the sugar beet samples (11.82%). This suggests that sugarcane has a higher initial ethanol concentration. For sugarcane, the ethanol concentration decreases sharply after the first fraction, with some variability, but generally follows a trend of diminishing ethanol content across subsequent fractions. However, its noticeable for fraction 5, the % yield was 12.32. For sugar beet, the ethanol concentration decreases more gradually across the fractions, from 11.82% to 0.07%. Sugar beet samples, while starting with lower ethanol concentrations, show a more gradual decrease in the % yield of ethanol.
Table 3.0 shows the average ethanol content per fractions of distillate collected. As can be seen, the average ethanol content was higher for sugar cane than sugar beet. Six fractions were collected from distillate of both samples and these were progressively lower in ethanol content as the more volatile ethanol content were distilled off. Figure 4 and Figure 5 show the GC profile for the sugar cane and sugar beet samples and these can be compared with the GC profile for the Standard, Figure 3. In addition to ethanol, as the main component, other components were observed from the GCs. These are shown in the Table 4.0. The prominent ethanol peak within the region, 4 to 4.4 is noticeable.
Based on the results, 530g of sugar cane juice will produce 10.18ml of bioethanol. Thus, in terms of scalability, 1 tonne (1.0 x 106g) of sugar cane juice will produce 19.21 litres of ethanol. On a parallel trend, 605g of beet will yield 4.103 ml of bioethanol. Thus, 1 tonne (1.0 x 106g) of beet will produce 6.781 litres of bioethanol. Thus, the sugar cane which is a sustainable feedstock in Guyana, can be grown for bioethanol production in addition to its processing to make sugar. Also, for the sugar cane and sugar beet, the Brix conversion ratio is 0.191 Brix per hour and 0.079 Brix per hour respectively.
Based on the ANOVA results, Table 5, there are no statistically significant differences in ethanol concentration between the sugarcane and sugar beet samples, across different fractions, or in the interaction between the type of sample and the fraction. The F-value for the sample effect is 2.03236, which is less than the critical F-value of 4.17. The P-value of 0.164304 is greater than the significance level of 0.05. The ANOVA analyses amongst the different fractions (1-6) for both substrates also revealed no statistically significant different. A P-value of 0.159 was noted, Table 6. When the ANOVA analyses were also applied to the two major fractions (1-2) for both substrates, a P-value of 0.47 was noted. This is shown in Table 6. Therefore, the hypothesis (H1) that either sugarcane or sugar beet is significantly better for ethanol production is not supported by these ANOVA analyses.
Limitations to research: The inability to conduct the GC analyses on the distillates at the University of Guyana. The student had to seek analyses at the Quality Assurance Department at Demerara Distillers Limited. This imposes a time and transportation constraints.
Conclusions
This study demonstrated that both sugarcane and sugar beet are viable feedstocks for bioethanol production, with each exhibiting unique fermentation profiles. Sugarcane showed a higher % ethanol concentrations of 10.18% and by-products compared to sugar beet (4.1%), indicating its potential for higher ethanol yields. In addition, sugar cane demonstrated a higher initial average Brix content of 18.97, compared to the average Brix content of 7.87 for beet fruit. This demonstrated that sugar cane has the potential to produce more bioethanol, compared with sugar beet. Guyana has one bioethanol plant to date, with a production capacity of 1000 litres per day. However, the abundance of sugar cane should allow for the establishment of more bioethanol plant around the country.
Acknowledgements
We thank the department of chemistry for bench space to conduct the above research. Also, the Quality Assurance Department at Demerara Distillers Limited, Diamond, Guyana for GC analyses of the distillate.
References
- Demirbas AH, Demirbas I (2007). “Importance of rural bioenergy for developing countries”. Energy Conversion Management. 48, 2386-2398; https://doi.org/10.1016/j.enconman.2007.03.005
- Demirbas A (2008). Biofuels sources, biofuel policy, biofuel economy and global biofuel projections. Energy Conversion and Management. 49, 2106-2116; https://doi.org/10.1016/j.enconman.2008.02.020
- Yu Z., Zhang H. (2004). Ethanol fermentation of acid-hydrolysed cellulosic pryolysate with Saccharomyces cerevisiae. Biores.Technol. 93, 199-204; https://doi: 10.1016/j.biortech.2003.09.016.
- Reddy VL, Reddy OVS (2007). Production of Ethanol from Mango (Mangifera indica I) fruit Juice Fermentation, Research Journal of Microbiology, 2(10): 763-769.
- Solomons GWT, Fryhle, CB, Organic Chemistry. (2008). 9th Edition, John Wiley and Sons, Inc.2008.
- Martin M, Galbe M, Wahlborn CF, Hahn-Hagerdal B, Jonsson LJ. (2002). “Ethanol production from enzymatic hydrolysates of sugarcane bagasse using recombinant xylose-utilising Saccharomyces cerevisiae. Enzyme Micro.Technol. 31: 274-282; https://doi.org/10.1016/S0141-0229(02)00112-6
- Graham, RW, Reynolds TW, Hsu Y. (1976). Preliminary assessment of systems for deriving liquid and gaseous fuels from waste or grown organics. US Department of Commerce, National Technical Information Service. 1-40.
- Dutta A, Mukherjee A. (2010). Comparison of alcohol production in batch culture using different substrates by Saccharomyces cerevisiae. Biomedical and Pharmacology Journal, 3(1), 23-26; http://biomedpharmajournal.org/?p=1120
- Reddy VL, Reddy OVS. (2009). Production, optimization and characterization of wine from Mango (Mangifera indica Linn Natural Product Radiance. 8(4): 426-435.
- Naik SN, Goud VV, Rout PK, Dalai AK. (2010). “Production of first and second generation biofuels: a comprehensive review, “Renewable and Sustainable Energy Reviews, 14 (2), 578-597; https://doi.org/10.1016/j.rser.2009.10.003
- Hossain ABMS, Ahmed SA, Ahmed MA, Adnan FMA, Annuar MSM, Mustafa H, Hammad N. (2011). “Bioethanol fuel production from rotten banana as an environmental waste management and sustainable energy, African Journal of Microbiology Research. 5(6), 586-598. https://doi.org/10.5897/AJMR10.231
- Ingale S, Joshi S, Gupte A. (2014). Production of bioethanol using agricultural waste: banana pseudo stem. Braz J. Microbiol. 45(3): 885–892; https://doi: 10.1590/S1517-83822014000300018
- Reddy V, Reddy OVS. (2009). Production, optimization and characterization of wine from Mango (Mangifera indica Linn Natural Product Radiance. 8(4), 426-435.
- Massengo V, Loumouamou BW, Diakabana P, Silou T. (2014). Ethanol production by fermentation of the pulp of the “BOKO” mango”. International Journal of Chemical Science and Technology. 4(4), 71-77.
- Wairagu NW, Kiptoo J, Githiomi JK. (2013). Nutritional assessment of Sclerocarya birrea (amarula) fruit from Kenya. International Journal of Current Research. 5(5):1074–1078.
- Chanprasartsuk O, Pheanudomkitlert K, Toonwai D. (2012). Pineapple wine fermentation with yeasts isolated from fruit as single and mixed starter cultures. As. J. Food Ag-Ind. 5(02), 104-111
- Mohammed S. et al, (2014). “Bioethanol Production from Mango Waste (Mangifera indica L.): Biomass as Renewable Energy”
- Chowdhury P, Ray R.C. (2007). Fermentation of Jamun (Syzgium cumini L.) Fruits to Form Red Wine. ASEAN Food Journal. 14 (1): 15-23.
- Patil SS, Thorat RM, Rajasekaran P. (2012). Fermentation of Jamun (Syzgium cumini L.) Fruits to Form Red Wine, Journal of Advanced Laboratory Research in Biology. 3(3): 234-238.
- Tropea A, Wilson D, Giovanna Loredana La Torre, Lo CURTO Rosario, Saugman P, Davies, PT. (2014). Bioethanol Production From Pineapple Wastes. Journal of Food Research; Published by Canadian Centre of Science and Education. 3 (4); 60-70.
- Mishra J. Kumar D, Samanta S, Vishwakarma M. (2012). “A comparative study of ethanol production from various agro residues by using Saccharomyces cerevisiae and Candida albicans. Journal of Yeast and Fungal Research. 3(2), 12 – 17. https://doi: 10.5897/JYFR11.025
- Khan Z, Dwivedi AK. (2013). Fermentation of Biomass for the production of ethanol: Universal Journal of Environmental Research and Technology. 3 (1), 1-13. (Alexica Brown References, from Below)
- Beigbeder J, De Medeiros Dantas JM, Lavoie J. (2021). Optimization of yeast, sugar and nutrient concentrations for high ethanol production rate using industrial sugar beet molasses and response surface methodology. Fermentation, 7(2), 86.; Crossreference
- Berłowska J, Pielech‐Przybylska K, Balcerek M, Cieciura W, Borowski S, Kręgiel D. (2017). Integrated Bioethanol Fermentation/Anaerobic Digestion for Valorization of Sugar Beet Pulp. Energies, 10(9), 1255 https://doi.org/10.3390/en10091255
- Berłowska J, Cieciura-Włoch W, Kalinowska H, Kręgiel D, Borowski S, Pawlikowska E, Binczarski M, Witońska, I. (2018). Enzymatic conversion of sugar beet pulp: a comparison of simultaneous saccharification and fermentation and separate hydrolysis and fermentation for lactic acid production. Food Technology and Biotechnology, 56(2). https://doi.org/10.17113/ftb.56.02.18.5390
- Brasil MA, Gomes LH, Kamogawa MY, Basso LC. (2020). Ethanol determination in fermented sugarcane substrates by a diffusive micro-distillation device. Journal of Microbiological Methods, 178, 106085. https://doi.org/10.1016/j.mimet.2020.106085
- Bušić A, Marđetko N, Kundas S, Morzak G., Belskaya H, Šantek MI, Komes D, Novak S, Šantek B. (2018). Bioethanol Production from Renew able Raw Materials and its Separation and Purification: a Review. Food Technology and Biotechnology, 56(3). https://doi.org/10.17113/ftb.56.03.18.5546
- Charnock H, Kemp B. (2022). Influence of cane and beet sugar for second fermentation on “fruity” aromas in Auxerrois sparkling wines. OENO One, 56(2). https://doi.org/10.20870/oeno-one.2022.56.2.4864
- Chaudhary A, Chand B, Kaur C. (2014.). Bioethanol production from fruit peels using simultaneous saccharification and fermentation. GSTF Journal of Biosciences, 3(1). https://doi.org/10.5176/2251-3140_3.1.51
- Clifford, T., Howatson, G., West, D. J., & Stevenson, E. (2015). The potential benefits of red beetroot supplementation in health and disease. Nutrients, 7(4), 2801–2822. https://doi.org/10.3390/nu7042801
- Dimidi, E., Cox, S., Rossi, M., & Whelan, K. (2019.). Fermented Foods: Definitions and Characteristics, Impact on the Gut Microbiota and Effects on Gastrointestinal Health and Disease. Nutrients, 11(8). https://doi.org/10.3390/nu11081806
- Duraisam R, Salelgn K, Berekete AK. (2017). Production of Beet Sugar and Bio-ethanol from Sugar beet and it Bagasse: A Review. International Journal of Engineering Trends and Technology, 43(4), 222–233. https://doi.org/10.14445/22315381/ijett-v43p237
- Eriklioglu H, Ilhan E, Khodasevich M, Korolko D, Manley M, Castillo R, Oztop MH. (2023). Classification and quantification of sucrose from sugar beet and sugarcane using optical spectroscopy and chemometrics. Journal of Food Science, 88(8), 3274–3286. https://doi.org/10.1111/1750-3841.16674
- Fasahat P, Aghaeezadeh M, Jabbari L, Hemayati S S, Townson P. (2018b). Sucrose accumulation in sugar beet: From fodder beet selection to genomic selection. Sugar Tech, 20(6), 635–644. https://doi.org/10.1007/s12355-018-0617-z
- Flores K, Neil S, Nguyen JJ, Capper K, Felder M. (2018). Sugar Rush: How Fermentation Rate Increases with Glucose Concentration. Flores | Journal of Undergraduate Biology Laboratory Investigations. https://undergradsciencejournals.okstate.edu/index.php/JUBLI/article/view/8522
- FreshPoint. (2022). Understanding Brix: Is a higher number always better? | FreshPoint. FreshPoint. https://www.freshpoint.com/news/understanding-brix-is-a-higher-number-always-better/
- Grabarczyk R, Urbaniec K, Wernik J, Trafczyński M. (2021). Evaluation of the Two-Stage Fermentative Hydrogen Production from Sugar Beet Molasses. Energies, 12(20), 4090. https://doi.org/10.3390/en12214090
- Gumienna M, Szambelan K, Jeleń HH, Czarnecki Z. (2014). Evaluation of ethanol fermentation parameters for bioethanol production from sugar beet pulp and juice. Journal of the Institute of Brewing, 120(40), 543-549. https://doi.org/10.1002/jib.181
- Hernández-Salas J, Ramírez V, Rendón VJS, Rivera-Hernández, KN, González-César R, Plascencia-Espinosa, MÁ, Trejo-Estrada, SR. (2009b). Comparative hydrolysis and fermentation of sugarcane and agave bagasse. Bioresource Technology, 100(3), 1238–1245. https://doi.org/10.1016/j.biortech.2006.09.062
- Hernawan MR, Pratiwi D, Wahono SK, Darsih C, Hayati S, Poeloengasih CD, Nisa K, Indrianingsih AW, Prasetyo DJ, Jatmiko TH, Kismurtono M, Rosyida VT. (2017). Bioethanol production from sugarcane bagasse by simultaneous sacarification and fermentation using Saccharomyces cerevisiae. Nucleation and Atmospheric Aerosols, 1823(1). https://doi.org/10.1063/1.4978099
- Imlimthan S, Figueiredo P, Santos HA, Sarparanta M. (2021). Introduction to lignocellulosic materials. In Elsevier eBooks (pp. 1–34). https://doi.org/10.1016/b978-0-12-820303-3.00010-2
- Jeswani HK, Chilvers A, Azapagic A. (2020). Environmental sustainability of biofuels: a review. Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences, 476(2243). https://doi.org/10.1098/rspa.2020.0351
- Khongchamnan P, Suriyachai N, Kreetachat T, Laosiripojana N, Weerasai K, Champreda, V, Suwannahong K, Sakulthaew C, Chokejaroenrat C, Imman S. (2022). Optimization of Liquid Hot Water Pretreatment and Fermentation for Ethanol Production from Sugarcane Bagasse Using Saccharomyces cerevisiae. Catalysts, 12(5) https://doi.org/10.3390/catal12050463
- Koval O, Олійнічук СТ, Lysak T. (2021). The comparative study on two commercial strains of saccharomyces cerevisiae for ethanol production from hard-to-ferment sugar-containing raw materials. Harčova Nauka Ì Tehnologìâ, 15(1). https://doi.org/10.15673/fst.v15i1.1960
- Leiper KA, Schlee, C., Tebble, I., & Stewart, G. G. (2006). The fermentation of beet sugar syrup to produce bioethanol. Journal of the Institute of Brewing, 112(2), 122–133. https://doi.org/10.1002/j.2050-0416.2006.tb00242.x
- Mannaa M, Han GS, Seo Y, Park I. (2021.). Evolution of Food Fermentation Processes and the Use of Multi-Omics in Deciphering the Roles of the Microbiota. Foods, 10(11), 2861. https://doi.org/10.3390/foods10112861
- Marzo C, Díaz AB, Caro I, Blandino A. (2019). Status and perspectives in bioethanol production from sugar beet. In Elsevier eBooks (pp. 61–79). https://doi.org/10.1016/b978-0-12-813766-6.00004-7
- Masson IDS, Costa GHG, Roviero JP, De Freita LA, Mutton MA, Mutton MJR. (2015). Bioethanol production from sweet sorghum broth and sugar cane fermentation. Ciencia Rural, 45(9), 1695–1700. https://doi.org/10.1590/0103-8478cr20130549
- Neves PV, Pitarelo AP, Ramos LP. (2016). Production of cellulosic ethanol from sugarcane bagasse by steam explosion: Effect of extractives content, acid catalysis and different fermentation technologies. Bioresource Technology, 208, 184–194. https://doi.org/10.1016/j.biortech.2016.02.085
- Rezić T, Oros D, Marković I, Kracher D, Ludwig R, Šantek B. (2013). Integrated hydrolyzation and fermentation of sugar beet pulp to bioethanol. Journal of Microbiology and Biotechnology, 23(9), 1244–1252. https://doi.org/10.4014/jmb.1210.10013
- Rolz C, De León R. (2011). Ethanol fermentation from sugarcane at different maturities. Industrial Crops and Products, 33(2), 333–337; https://doi.org/10.1016/j.indcrop.2010.11.013
- Roukas T, Kotzekidou P. (2020). Rotary biofilm reactor: A new tool for long-term bioethanol production from non-sterilized beet molasses by Saccharomyces cerevisiae in repeated-batch fermentation. Journal of Cleaner Production, 257, 120519. https://doi.org/10.1016/j.jclepro.2020.120519
- Sánchez C, Santos S, Sánchez R, Lienemann C, Todolí J. (2020). Profiling of organic compounds in bioethanol samples of different nature and the related fractions. ACS Omega, 5(33), 20912–20921. https://doi.org/10.1021/acsomega.0c02360
- Singh S, Tripathi AD, Chauhan AK, Gupta AK. (2021). Production of beetroot (Beta vulgaris L.) wine using different Saccharomyces strains and study of physicochemical and sensorial characteristics. Journal of Food Science and Technology, 58(11), 4442–4449. https://doi.org/10.1007/s13197-021-05136-y
- Siracusa V, Blanco I. (2020). Bio-Polyethylene (Bio-PE), Bio-Polypropylene (Bio-PP) and Bio-Poly(ethylene terephthalate) (Bio-PET): Recent Developments in Bio-Based Polymers Analogous to Petroleum-Derived Ones for Packaging and Engineering Applications. Polymers, 12(8), 1641. https://doi.org/10.3390/polym12081641
- Taveira, IC, Nogueira KMV, De Oliveira DLG, Silva RDN. (2021). Fermentation: humanity’s oldest biotechnological tool. Frontiers for Young Minds, 9. https://doi.org/10.3389/frym.2021.568656
- Tzeng D, Tai C, Chia Y, Lin Y, Ou AS. (2009). Investigation of fermenting conditions for sugarcane (saccharum officinaruml.) wine using response surface methodology. Journal of Food Processing and Preservation, 33(3), 330–346. https://doi.org/10.1111/j.1745-4549.2008.00254.x
- Vučurović VM, Puškaš V, Miljić U. (2018). Bioethanol production from sugar beet molasses and thick juice by free and immobilisedSaccharomyces cerevisiae. Journal of the Institute of Brewing, 125(1), 134–142. https://doi.org/10.1002/jib.536
- Yalcin SK, Ozbas ZY. (2008). Effects of pH and temperature on growth and glycerol production kinetics of two indigenous wine strains of Saccharomyces cerevisiae from Turkey. PubMed. https://doi.org/10.1590/s1517-838220080002000024
- Yücel HG, Aksu Z. (2015). Ethanol fermentation characteristics of Pichia stipitis yeast from sugar beet pulp hydrolysate: Use of new detoxification methods. Fuel, 158, 793–799. https://doi.org/10.1016/j.fuel.2015.06.016
- Yoon KY, Woodams EE, Hang YD. (2005). Fermentation of beet juice by beneficial lactic acid bacteria. LWT, 38(1), 73–75. https://doi.org/10.1016/j.lwt.2004.04.008
- Zaghloul RA, Ismail SA, Enan G, El-Meihy RM, Abdelrahman H. (2021). Maximization of Bio-Ethanol Production by Yeasts using Sugar Cane and Sugar Beet Molasses. Advances in Animal and Veterinary Sciences, 19(12), 2069-2076. https://doi.org/10.17582/journal.aavs/2021/9.12.2069.2076
- Technology Networks. (2024, March 12). Gas Chromatography – How a gas chromatography machine works, how to read a chromatograph and GCXGC. Analysis & Separations From Technology Networks. https://www.technologynetworks.com/analysis/articles/gas-chromatography-how-a-gas-chromatography-machine-works-how-to-read-a-chromatograph-and-gcxgc-335168